|
They will be grouped into two cohorts: individuals 18 through 64 years old and those aged 65 and older, with a goal of enrolling at least 25% of all volunteers who are 65 years old or older. Volunteers will be asked to give informed consent prior to their participation in the trial. The CoVPN includes existing NIAID-supported clinical research networks with infectious disease expertise and was designed for rapid and thorough evaluation of vaccine candidates and monoclonal antibodies for preventing COVID-19. trial sites participating are part of the NIAID-supported COVID-19 Prevention Network (CoVPN). The trial is being conducted in collaboration with Operation Warp Speed (OWS), a multi-agency collaboration overseen by HHS and the Department of Defense that aims to accelerate development, manufacture and distribution of medical countermeasures for COVID-19. “The launch of this study - the fifth investigational COVID-19 vaccine candidate to be tested in a Phase 3 trial in the United States - demonstrates our resolve to end the pandemic through development of multiple safe and effective vaccines.” “Addressing the unprecedented health crisis of COVID-19 has required extraordinary efforts on the part of government, academia, industry and the community,” said NIAID Director Anthony S. Department of Health and Human Services Office of the Assistant Secretary for Preparedness and Response, are funding the trial. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, and the Biomedical Advanced Research and Development Authority (BARDA), part of the U.S. Novavax is leading the trial as the regulatory sponsor. It will evaluate the safety and efficacy of NVX-CoV2373, a vaccine candidate developed by Novavax, Inc., of Gaithersburg, Maryland. The randomized, placebo-controlled trial will enroll approximately 30,000 people at approximately 115 sites in the United States and Mexico. The Phase 3 trial of another investigational coronavirus disease 2019 (COVID-19) vaccine has begun enrolling adult volunteers. 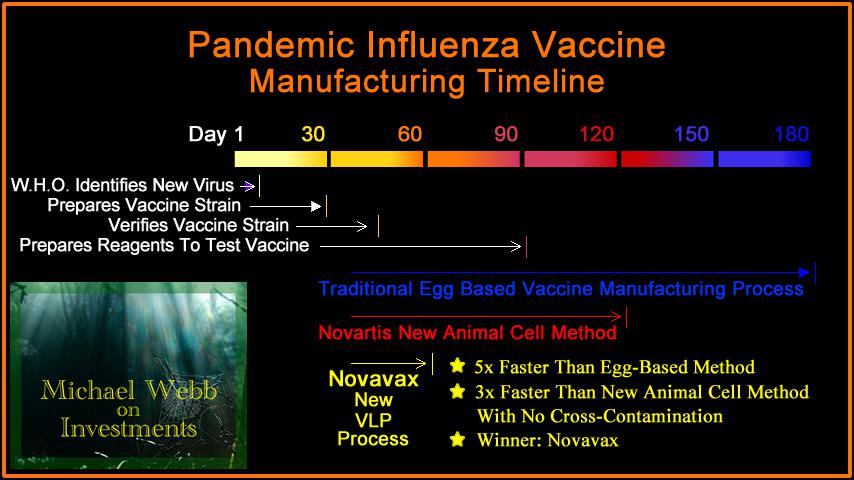 
Colorized scanning electron micrograph of an apoptotic cell (blue) infected with SARS-COV-2 virus particles (red), isolated from a patient sample.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
